PediatrOS ™ FlexTack ™ – For varus / valgus deformity
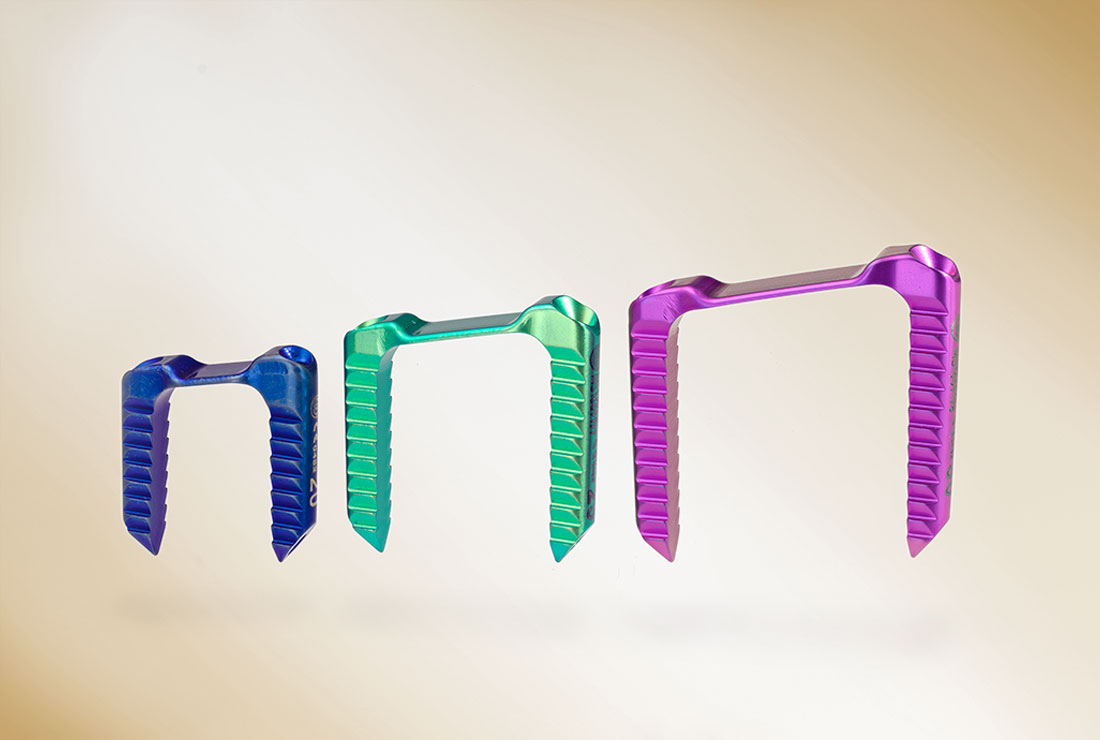
PediatrOS ™ FlexTack ™ – For varus / valgus deformity
PediatrOS™ FlexTack™
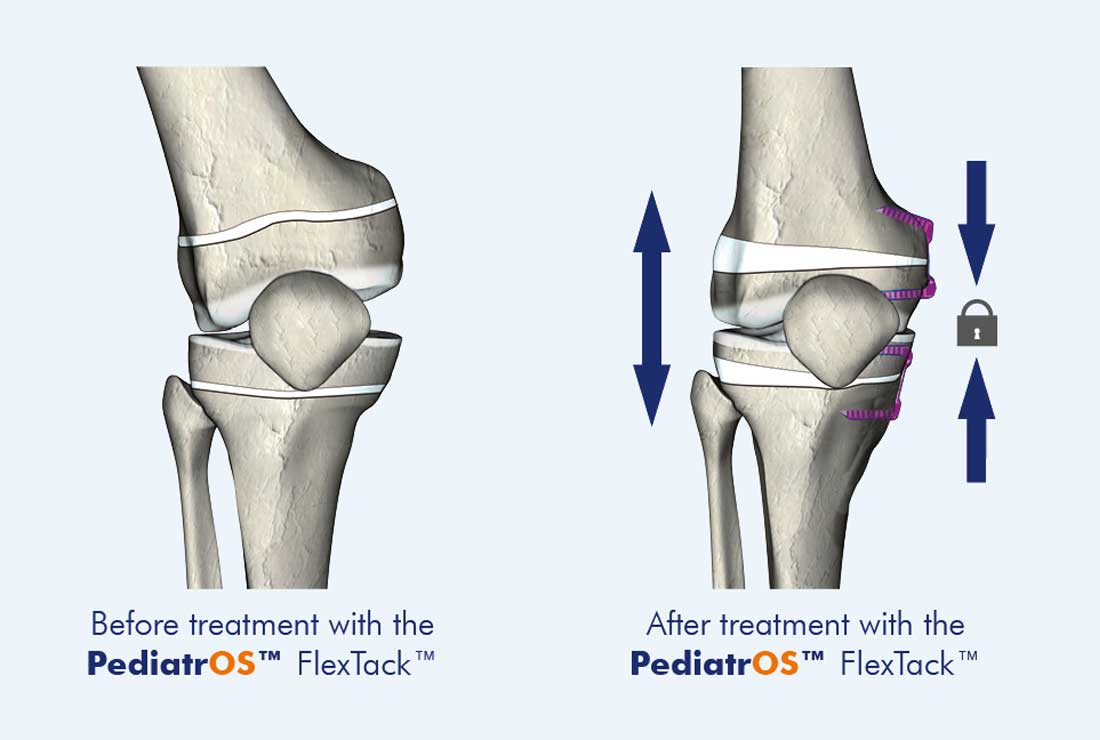
Growth guidance with varus/valgus misalignments
For correcting axis misalignment (Genu varum/valgum) using hemi-epiphysiodesis. The flexible center area of the anatomically shaped PediatrOS™ FlexTack™ allows it to bend open in vivo in response to bone growth forces, helping steer pediatric and adolescent bone growth gently and precisely. The trapezoidal design of the PediatrOS™ FlexTack™ staple is closely aligned to the anatomy of the femur and tibia. Cannulated staple legs allow precise placement using K-wires. This technological improvement on conventional implants for correcting varus/valgus misalignments reduces intraoperative fluoroscopy times and can be implanted using minimally invasive techniques.
Technical Data
- Trapezoidal design
- 3 sizes: 20 mm (blue), 25 mm (green) and 30 mm (purple)
- Barbed staple legs
- Cannulated staple legs to facilitate insertion over 1.6 mm K-wires for optimal intraoperative alignment and post-operative growth guidance
Features
- First anatomically shaped titanium implant designed to correct angular deformities
- Flexible staple bridge providing a bending zone that improves correction rates through an extra-epiphyseal fulcrum. Reduced implant-associated complication rates compared to two-hole plates. No fracturing screws in large, long-lasting corrections
- Supplied sterile
- Minimal invasive surgery with reduced overall OR-time. Achieved by reducing the number of implants from two screws and a compression plate to a single staple per epiphysiodesis site
- Ideal biomechanical alignment
- Fluoroscopy time reduction of 35 % compared to plating systems. Consequently, reduced x-ray exposure for patients
- Alignment template ensures precise placement
- Easy to remove using 2.0 mm K-wire with threaded tip
- ONE set of instruments for two indications: PediatrOS™ FlexTack™ and PediatrOS™ RigidTack™
- Immediate weight-bearing possible
Indications
Specific pediatric conditions/diseases for which the devices is indicated include:
- Valgus, varus, or flexion, extension, deformities of the knee (femur and/or tibia)
- Valgus or varus deformities of the elbow (humerus)



